Compound (2)
Article Title (3)
Assay (3)
Amundsen, R; �sberg, A; Ohm, IK; Christensen, H Cyclosporine A- and tacrolimus-mediated inhibition of CYP3A4 and CYP3A5 in vitro. Drug Metab Dispos 40: 655 -61 (2012) Liu, Y; Ruan, H; Li, Y; Sun, G; Liu, X; He, W; Mao, F; He, M; Yan, L; Zhong, G; Yan, H; Li, W; Zhang, Z Potent and Specific Inhibition of NTCP-Mediated HBV/HDV Infection and Substrate Transporting by a Novel, Oral-Available Cyclosporine A Analogue. J Med Chem 64: 543 -565 (2021) Hopkins, S; Scorneaux, B; Huang, Z; Murray, MG; Wring, S; Smitley, C; Harris, R; Erdmann, F; Fischer, G; Ribeill, Y SCY-635, a novel nonimmunosuppressive analog of cyclosporine that exhibits potent inhibition of hepatitis C virus RNA replication in vitro. Antimicrob Agents Chemother 54: 660 -72 (2010)
ChEMBL_1863252 (CHEMBL4364108) Displacement of fluorescein labelled cyclosporine A derivative from human recombinant CypD (30 to 207 residues) expressed in Escherichia coli cells incubated for 90 mins by fluorescence polarization assay Human ELISA assay Human peripheral blood mononuclear cells (PBMCs) are prepared from heparinized human blood by separation over a Ficoll density gradient.PBMCs are stimulated with phytohemagglutinin (PHA) in the presence of varying concentrations of compounds of the invention or cyclosporine A (CsA), a known inhibitor of cytokine production. Cytokine production is measured using commercially available human ELISA assay kits (from Cell Science, Inc.) following the manufacturers instructions.Alternatively, PBMCs with 10% FCS at 1-2�106/mL are stimulated with pre-coated with anti-CD3 (clone UCHT1) and anti-CD28 (clone ANC28.1/5D10) at 5 μg/mL each, with or without compound or DMSO (maximun concentration: 0.1%). Cell cultures are incubated at 37� C., 5% CO2. Samples of the culture supernatant are collected after 48-72 hrs. incubation for measurement of multiple cytokines. Cytokines present in the supernatants are quantified using BioRad BioPlex assays according to the manufacturer's instructions. GILZ gene assays HUT78 cells were cultured in IMEM plus 20% heat inactivated FBS and cell density was maintained between 0.1 to 1.2 million/mL. 786-O cells were cultured in RPMI plus 10% heat inactivated FBS. Actively growing cells were harvested and resuspended in HBSS with 2% FBS at 1.1 million cells per mL then dispensed to 384-well V-bottom plates at 45 μL per well. Serially diluted ADC solution was added to the cell plate (5 μL per well) and mixed for 2 min. Cells were then cultured at 37� C., at 5% CO2 for a designated time before supernatant was removed. Cells were harvested in lysis buffer from the Cells-to-Ct kit (40 μL, Life Technologies, 4391851C) following the supplier's protocol and mixed for 10 min followed by addition of 5 μL per well of stop solution from the kit. cDNA was synthesized with a reverse transcription kit (Life Technologies, 4391852C) follow by qPCR using the TaqMan gene expression master mix (Life Technologies, 4369016) with GILZ gene assay (Life Technologies, Hs00608272_m1) and GAPDH assay (Hs2758991_g1) in a duplex format with 3-4 technical replicates.Determination of apparent permeability was as follows. MDCKII cells (kindly provided by the Netherlands Cancer Institute, under a licensing agreement) were seeded on to 96-well transwell culture plates (Millipore Corp, Billerica, Mass.) and used in experiments after five days in culture. Test compound (1 μM) was prepared in Hank's Balanced Salt Solution (HBSS), 10 mM (4-(2-hydroxyethyl)-1-piperrazineethanesulfonic acid) (HEPES, pH 7.4), with 10 μM cyclosporine A (to inhibit endogenous transport) and 1.2 μM dextran Texas red (to confirm monolayer integrity). Substrate solution (150 μL) was added to either the apical (A) or the basolateral (B) compartment of the culture plate, and buffer (150 μL; HBSS, 10 mM HEPES, pH 7.4) with 10 μM cyclosporine A was added to the compartment opposite to that containing the substrate. At t=3 hr, 50 μL samples were removed from both sides of monolayers dosed with test compound and placed in 96 well plates, 50 μL internal standard (1 μM labetolol) and 100 μL HBSS was added to the samples. Samples were analyzed by LC/MS/MS using an Applied Biosystems SCIEX API 5000 triple quadruple mass spectrometer (Concord, ON, Canada) with a TurbolonSpray ion source in the positive ion mode. A Thermo Scientific Transcend LX-2 system (Franklin, Mass.) was coupled to the API 5000 with a flow rate of 800 μL/min to direct sample into the mass spectrometer.
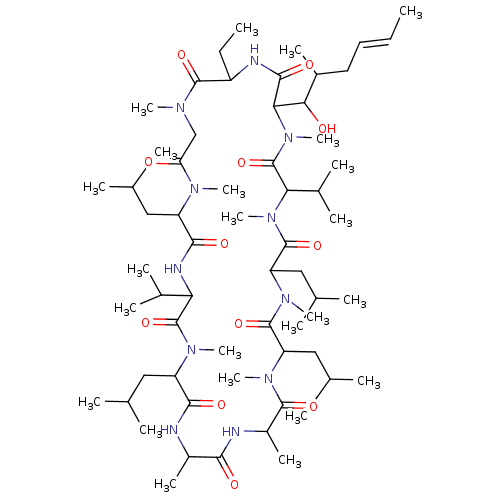 US11802108, Compound CSA US9090657, Cyclosporine A, 1 BDBM172718
US11802108, Compound CSA US9090657, Cyclosporine A, 1 BDBM172718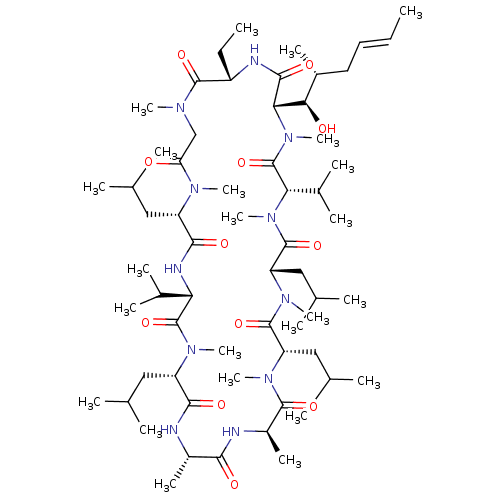 30-Ethyl-33-((E)-1-hydroxy-2-methyl-hex-4-enyl)-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,28-octamethyl-1,4,7,10,13,16,19,22,25,28,31undecaaza-cyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone (3S,6S,9S,12R,15S,18S,21S,24S,30S,33S)-30-ethyl-33-[(1R,2R,4E)-1-hydroxy-2-methylhex-4-en-1-yl]-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-bis(propan-2-yl)-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecone Cyclosporine A US10077289, Compound Cyclosporin A US9138393, Cyclosporin A 30-Ethyl-33-(1-hydroxy-2-methyl-hex-4-enyl)-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31undecaaza-cyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone Cyclosproine A Cyclosporin A BDBM50022815 CYCLOSPORINE
30-Ethyl-33-((E)-1-hydroxy-2-methyl-hex-4-enyl)-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,28-octamethyl-1,4,7,10,13,16,19,22,25,28,31undecaaza-cyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone (3S,6S,9S,12R,15S,18S,21S,24S,30S,33S)-30-ethyl-33-[(1R,2R,4E)-1-hydroxy-2-methylhex-4-en-1-yl]-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-bis(propan-2-yl)-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecone Cyclosporine A US10077289, Compound Cyclosporin A US9138393, Cyclosporin A 30-Ethyl-33-(1-hydroxy-2-methyl-hex-4-enyl)-6,9,18,24-tetraisobutyl-3,21-diisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-1,4,7,10,13,16,19,22,25,28,31undecaaza-cyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone Cyclosproine A Cyclosporin A BDBM50022815 CYCLOSPORINE